Laser Marking for Medical Devices: Why Traceability Is Non‑Negotiable
In today’s medical device manufacturing landscape, traceability isn’t just a tick-box exercise, it’s an absolute necessity rooted in both law and quality assurance. Devices ranging from intricate surgical instruments to simple single-use diagnostics must remain identifiable and traceable at every step of their lifecycle. From manufacture and sterilisation, through distribution and clinical use, to potential recall or end-of-life management, traceability ensures accountability and, most importantly, patient safety.
This regulatory focus on traceability isn’t just about preventing mistakes or recalls; it’s about building an environment where every device can be unambiguously identified and accounted for, instilling confidence for patients, healthcare professionals, and regulators alike.

Laser Marking: The Ultimate Medical Device Manufacturing UDI Solution
The Regulatory Landscape in the UK
Since Brexit, the regulatory environment for medical devices in the UK has undergone significant transformation. While the backbone remains the Medical Devices Regulations 2002 (UK MDR 2002), the Medicines and Healthcare products Regulatory Agency (MHRA) continues to update and evolve the regime, increasing alignment with global best practice and introducing requirements similar to those found in the European Union’s Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR).
A core element that persists, and is only becoming stricter, is the requirement for robust traceability. For devices placed on the UK market, clear, durable, and permanent marking is more than just good practice, it’s fast becoming a legal requirement. It’s especially critical for reusable devices, which must withstand rigorous cleaning, sterilisation, and handling. Temporary solutions like inkjet, labels or simple mechanical engraving frequently fail these demanding standards, particularly in clinical and sterile environments where mark degradation can directly impact patient safety and recall effectiveness.
The Shift to Unique Device Identification (UDI)
The UK is steadily introducing a UDI system similar to that used in the EU and US. UDI involves marking medical devices with a machine-readable and human-readable code that uniquely identifies the product down to batch, lot, or serial number. While some provisions are still being phased in, regulators and notified bodies are increasingly expecting UDI direct marking, especially on reusable or higher-risk devices, wherever technically feasible. This is part of a broader international movement towards harmonised device identification to improve safety and streamline post-market surveillance.
The Irish and EU Regulatory Environment
If your business is based in Ireland, operates across the EU, or exports to the EU from the UK, the requirements are even more explicit. The Republic of Ireland adheres strictly to the EU Medical Device Regulation (MDR 2017/745) and In Vitro Diagnostic Regulation (IVDR 2017/746), both among the world’s most stringent frameworks for device traceability.
These regulations require:
- Mandatory UDI System: All medical devices, especially reusable/high-risk categories, must feature a direct part marking of the UDI where technically feasible.
- Permanence & Legibility: The mark must withstand the entire service life of the device, through all cleaning, sterilisation, and daily use, without degrading.
- Enforcement & Market Access: Irish manufacturers are regulated by the Health Products Regulatory Authority (HPRA) and are subject to EU-wide post-market surveillance and recall coordination, in which traceability is central. Without permanent, machine-readable, and legible marking, non-compliance puts both approvals and market access at risk.
Laser marking is typically the only marking technology that can fully satisfy these legal, technical, and hygiene demands. For Irish and EU-based manufacturers, adopting repeatable, validated laser marking is indispensable, not just for regulatory compliance, but also for securing international distribution and maintaining reputational trust.
Why Laser Marking Sets the Bar for Compliance
Amid these regulatory demands, laser marking has emerged as the clear industrial standard for medical device identification. Here’s why:
- Unmatched Permanence: Laser marking directly modifies the material surface, be it metal, plastic, or ceramic, producing marks that are resistant to wear, aggressive cleaning, sterilisation, and repeated handling. Unlike ink, adhesives or labels, laser marks don’t fade, peel, or contaminate sensitive environments.
- Sterility and Cleanroom Compatibility: No chemicals, inks, or consumables means zero risk of introducing contaminants. Laser marking is inherently suitable for cleanroom environments and supports the strict hygiene controls of medical manufacturing.
- Technical Versatility: Lasers, especially those with advanced optics and 3-axis control, can mark flat, curved, recessed, or ultra-small surfaces. This versatility is indispensable for today’s medical devices, which may offer limited area for identification or possess complex geometries.
- Corrosion-Resistant Identification: On metals such as stainless steel, laser annealing creates dark, high-contrast marks by altering the metal’s oxide layer without removing material, ensuring identifiers that are readable, non-contaminating, and remain corrosion-resistant throughout the instrument’s life.
- Materials Flexibility: Whether it’s PE, PEEK, or advanced polymer blends, fibre and UV lasers create highly legible, permanent, and non-contaminating marks on plastics. This flexibility is critical for the diversity of materials now used in diagnostics, disposables, and implants.
- Integrated Compliance Features: Modern laser marking systems, such as those from FOBA, incorporate integrated vision cameras for automatic code alignment, verification, and immediate defect detection. This supports ISO 13485, Good Manufacturing Practice (GMP), and ISO 14971 requirements for risk management and documentation.
Common Applications in UK Medical Manufacturing
In all these cases, laser technology helps UK manufacturers achieve permanent, legible, and compliant identification without introducing new risks or delays into the process. The technology is compatible with automation and cleanroom integration, helping to future-proof production lines for evolving standards.
In the UK, laser marking is used extensively across orthopaedics, diagnostics, surgical instrumentation, and dental product manufacturing. Implants such as hip stems or trauma plates are engraved with lot numbers, expiry dates and manufacturer codes. Surgical tools are laser annealed for traceability while maintaining resistance to rust and surface degradation. Diagnostic test components and housings are marked with batch codes that remain legible throughout logistics and clinical use.


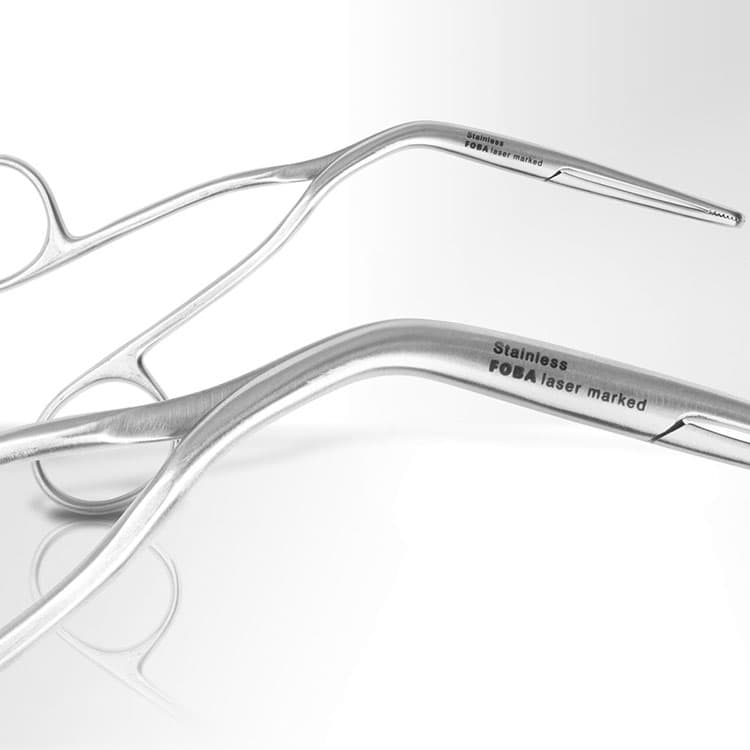
Meeting UDI Requirements: A Technological Perspective
In practice, UDI marking must be both human-readable and machine-readable, typically as a DataMatrix code alongside alphanumeric text. For Class II and III reusable devices, regulators in both the UK and EU expect UDIs to be marked directly onto the device; labels or packaging alone are not sufficient.
Laser marking excels in this context. FOBA’s high-precision fibre and UV laser systems are designed to generate flawless, high-contrast DataMatrix and alphanumeric codes readable by both humans and barcode scanners vital for digital traceability systems. Integrated software and camera systems further ensure each device is correctly marked and that the UDI meets international standards for readability and permanency. This automated, closed-loop approach significantly reduces the risk of marking errors and supports full audit trails.
FOBA & TLM Laser: Partners in Compliance Across the UK and Ireland
FOBA’s medical-grade laser marking systems are benchmark solutions for regulatory compliance. Whether you need a turnkey table top machine for small batches or an inline system for high-speed automation, FOBA’s technology, from advanced fibre lasers to UV and CO₂ options, can be customised for your specific material, geometry, and throughput needs.
- Proprietary Marking Software: From UDI data entry and validation to automated vision-based code verification and process logging.
- Zero Defect Manufacturing: Camera-based alignment detects and rejects mis-marked parts in real time, supporting your ISO 13485-compliant quality system.
- Flexible Integration: FOBA’s modular designs suit both fully automated lines and smaller, specialist facilities.
As the official FOBA distribution partner for both the UK and Ireland, TLM Laser offers comprehensive services, from feasibility assessment and application testing, installation, training, and after-sales maintenance support. We guide manufacturers through every step, ensuring that your marking solution satisfies not just today’s regulatory demands, but those of tomorrow as well.
Rethink Traceability for the Future
Continuing with ink, labels, or contact-based methods? As UK and EU regulations rapidly evolve, legacy marking can result in compliance gaps or even market exclusion. With FOBA laser marking, you achieve clean, permanent, and fully traceable identification, perfectly aligned with the demands of regulators, auditors, and the global supply chain.
Preparing for an audit, reviewing your UDI strategy, or simply future-proofing your traceability process? Contact TLM Laser today for expert advice, hands-on demonstrations, or a sample marking trial and see how we can support your compliance journey with FOBA laser solutions.

Talk to Us!
Connect with our Technical & Support Team.


